Mar 19, 2026
Treatment-Resistant Depression: Augmentation and Advanced Therapies

Confused by multiple generic options? Learn how to use the FDA Orange Book and bioequivalence ratings to choose the safest, most effective generic medication.

Generics share active ingredients with brands, making interactions nearly identical. Learn about narrow therapeutic index exceptions, excipient sensitivities, and how to monitor safety.
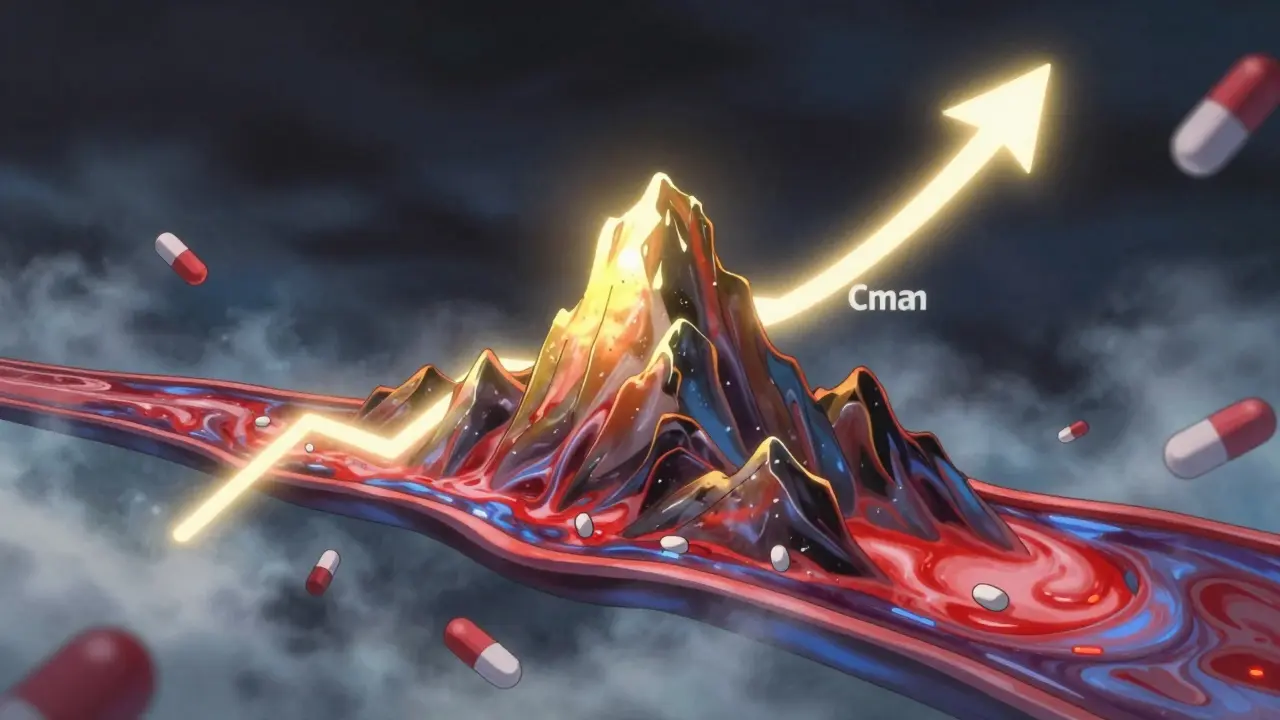
Cmax and AUC are the two key pharmacokinetic measures used to prove generic drugs work like brand-name versions. Cmax shows peak concentration, AUC shows total exposure-both must fall within 80%-125% for approval.
